 
Scientists also use trends in the periodic table to predict reactivity of metals and non-metals. The series is basically a chart listing elements according to how readily the metals displace H2 in reactions. The metal activity series can help scientists predict which metal will be the most reactive and to compare reactivity of different metals. That makes them more reactive than other metals. They are ultra-keen to pass on this unwanted passenger to another element via bonding. But the alkali metals have only one electron on their outer shell. To achieve that feat, metals tend to shed electrons. 
Or in other words, they strive to have a full outer electron shell. Generally speaking, elements strive to be chemically stable. Some of these metals can produce explosions when dropped in water. 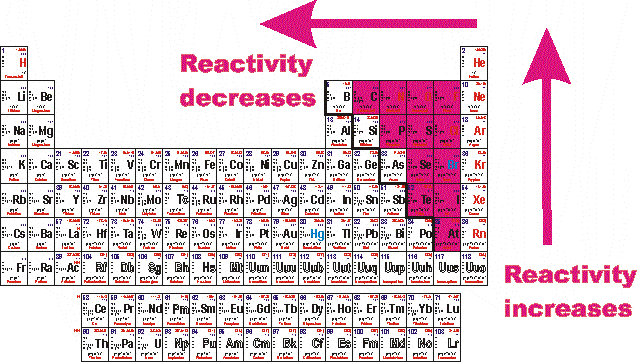
Did you know that some metals are so reactive that they explode on contact with water? Certain metals like potassium, sodium, lithium, rubidium, and more, are so reactive, that they oxidize (or tarnish) instantly when exposed to air. The following chart summarizes the reactions discussed above. Iron will react with sulfuric acid to give hydrogen and iron (II) sulfate.įe (s) + H 2SO 4 (l) → FeSO 4 (aq.) + H 2 (g) Zinc (Zn) reacts with sulfuric acid (H 2SO 4) to produce zinc sulfate (ZnSO 4) while liberating hydrogen gas (H 2). Metals that lose electrons easily produce a fast and vigorous reaction.ġ. The reactivity of the metal determines the reaction rate. The amount of hydrogen produced depends on the starting mass of the metal and the acid’s strength. The electrons and protons combine to form a hydrogen atom. Each metal atom loses one or more electrons, and each acid molecule loses one or more protons. Highly reactive metals produce hydrogen when reacting with acid. Aluminum (Al) reacts vigorously with water to produce hydrogen gas.Ģ Al (s) + 6 H 2O (l) → 2 Al(OH) 3 (aq.) + 3 H 2 (g) 3. Sodium (Na) is highly reactive and displaces hydrogen (H 2) from water (H 2O).Ģ Na (s) + 2 H 2O (l) → 2 NaOH (aq.) + H 2 (g)Ģ. Highly reactive metals can react with cold water to form the corresponding hydroxide.ġ. Magnesium (Mg) can displace copper (Cu) in an aqueous solution of copper nitrate (Cu(NO 3) 2). Copper (Cu) can displace silver (Ag) in an aqueous solution of silver nitrate (AgNO 3).Ĭu (s) + 2 AgNO 3 (aq.) → Cu(NO 3) 2 (aq.) + 2 Ag (s/ppt.)Ģ. A characteristic of this reaction is that one cation trades places with another to form a new product.ġ. This reaction is known as a single-displacement reaction. Metals higher in the series tend to lose electrons and easily displace lower metals. 
Based on the importance of the reactivity series, let us look at the various chemical reactions that metals undergo. It helps us understand why different metals are extracted from their ores differently. Aside, it can predict the outcome of single-displacement reactions. It can predict the reactions between metals and water and metals and acids. The reactivity series is essential for several reasons. įrom this list, it is clear that potassium is the most reactive metal. The list below represents the order of the reactivity series from most reactive to least reactive.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
